Free available chlorine. What is Free Available Chlorine? 2019-03-15
Grams per Liter of Available Chlorine

Small quantities of elemental chlorine are generated by oxidation of chloride to in as part of the immune response against bacteria. This term was later used as a generic term to describe all the elements in the chlorine family fluorine, bromine, iodine , after a suggestion by in 1826. Archived from the original on 2016-04-23. These compounds produced low levels of elemental chlorine and could be more efficiently transported than sodium hypochlorite, which remained as dilute solutions because when purified to eliminate water, it became a dangerously powerful and unstable oxidizer. They cannot keep a pool safe on their own. In the form of chloride , chlorine is necessary to all known species of life.
Free Available Chlorine

If it's cloudy, a little green, a serious dos … e of shock may be enough to stabilize it. Regular chlorination will ensure all harmful bacteria that pose some health problems in humans are eliminated. Labarraque's research resulted in the use of chlorides and hypochlorites of lime and of sodium in the boyauderies. In the subsurface environment, by 40 becomes more important as a way to generate 36Cl. Note: If adding Liquid Shock, also add 12 ounces of acid to counter balance the high pH. All four stable halogens experience intermolecular of attraction, and their strength increases together with the number of electrons among all homonuclear diatomic halogen molecules.
Free and combined chlorine
For an English translation of the relevant passages of this article, see: The Early History of Chlorine : Papers by Carl Wilhelm Scheele 1774 , C. Coughing and vomiting may occur at 30 ppm and lung damage at 60 ppm. You can help Wikipedia by. The most common compound of chlorine, common salt , has been known since ancient times. The concentration of chlorine required to successfully treat public water sources is high enough to be lethal to your fish. Even those that claim they do, aren't always fully effective in getting the job done, so choose your water treatment products carefully.
What is a Residual Chlorine?

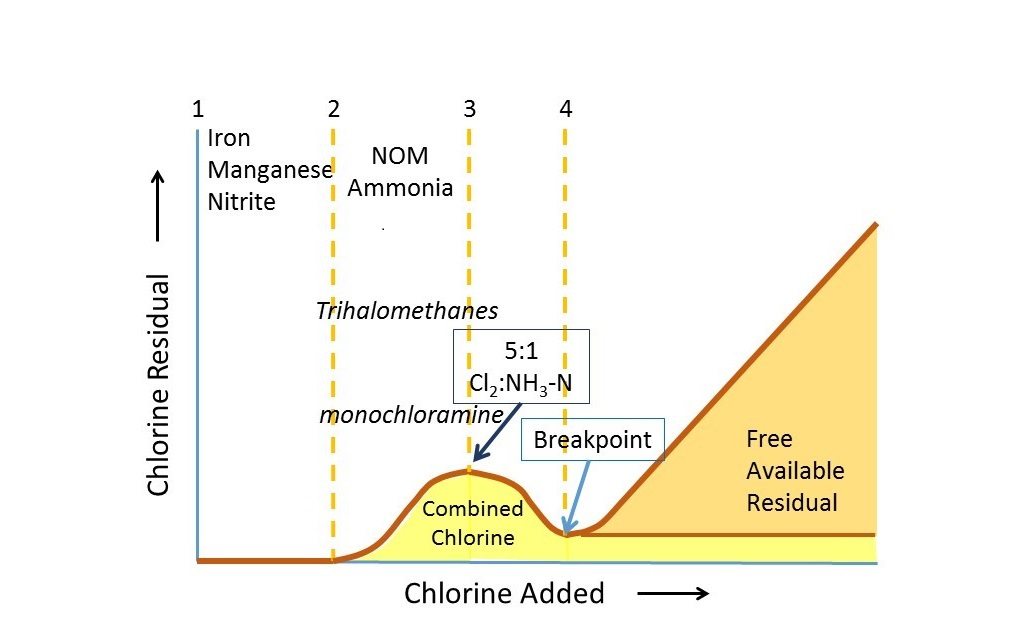
Total chlorine should be no more than 0. Read below if you want to learn more or just today. Like solid bromine and iodine, solid chlorine crystallises in the , in a layered lattice of Cl 2 molecules. Membrane cell process for chloralkali production In the , hydrogen chloride recovered from the production of is recovered as chlorine. When using borates, the recommended level is between 30 and 50 ppm. So, the Combined Chlorine the difference between the two tests is. If the Total Chlorine in your pool is higher than the Free Chlorine reading, then the difference between the two represents the level of Combined Chlorine in the water.
Easy Ways to Lower Chlorine in a Pool
The second-lightest of the , it appears between and in the periodic table and its properties are mostly intermediate between them. Direct contact with pool chlorine byproducts or off-gas can cause burns or skin blistering. Chlorine in water may be present in two forms, free and combined. About 15,000 chlorine compounds are used commercially. Fortunately, it is easily neutralized by one of two methods. This level will typically around 3,000, but different models vary.
Percent active chlorine
Free or residual chlorine is chlorine that is present in the form of hypochlorous acid, hypochlorite ions or as dissolved elemental chlorine. The mixing chamber contains a gold measurement electrode and a copper reference electrode. If you use any stabilizer in your swimming pool, then you should add chlorine to your swimming pool often so that you can keep the right amount of ppm concentration. John Snow to disinfect water from the cholera-contaminated well that was feeding the Broad Street pump in 1854 London, though three other reputable sources that describe that famous cholera epidemic do not mention the incident. You can prevent this from happening by keeping the water saturated with calcium. Along with chlorine, the method yields gas and , which is the most valuable product. Reduction in blood chloride leads to cerebral dehydration; symptoms are most often caused by rapid rehydration which results in.
What is a Residual Chlorine?

First and foremost, it means the old trick of aging water in open buckets or in a tank with a filter running, won't work anymore. Phosphate Phosphates are sometimes removed from the pool as a way of keeping algae in check. Better yet, use a test kit to get an accurate read of the chlorine, as described below. Its importance in food was very well known in and was sometimes used as payment for services for Roman generals and military tribunes. It is a strong oxidising agent, reacting with , , phosphorus halides, and. The Strange Case of the Broad Street Pump: John Snow and the Mystery of Cholera. Hyperchloremia can affect oxygen transport.
Free Available Chlorine in Water

If chlorine levels do not decrease within a couple days, try one of the methods below. The right response is usually to raise chlorine with a. As a disinfectant in water, chlorine is more than three times as effective against as , and more than six times as effective as. Chlorine-iron fire The element can combine with chlorine at high temperatures in a strong exothermic reaction, creating a chlorine-iron fire. Bound chlorine is chlorine that has been used up. Saturated sodium or potassium chloride solution is passed through the anode compartment, leaving at a lower. If the chlorine levels are only slightly high about 4—5ppm , chemicals are usually not necessary.