Define prothrombin activator enzyme. Blood: Hemostasis and Blood Coagulation Flashcards 2019-02-05
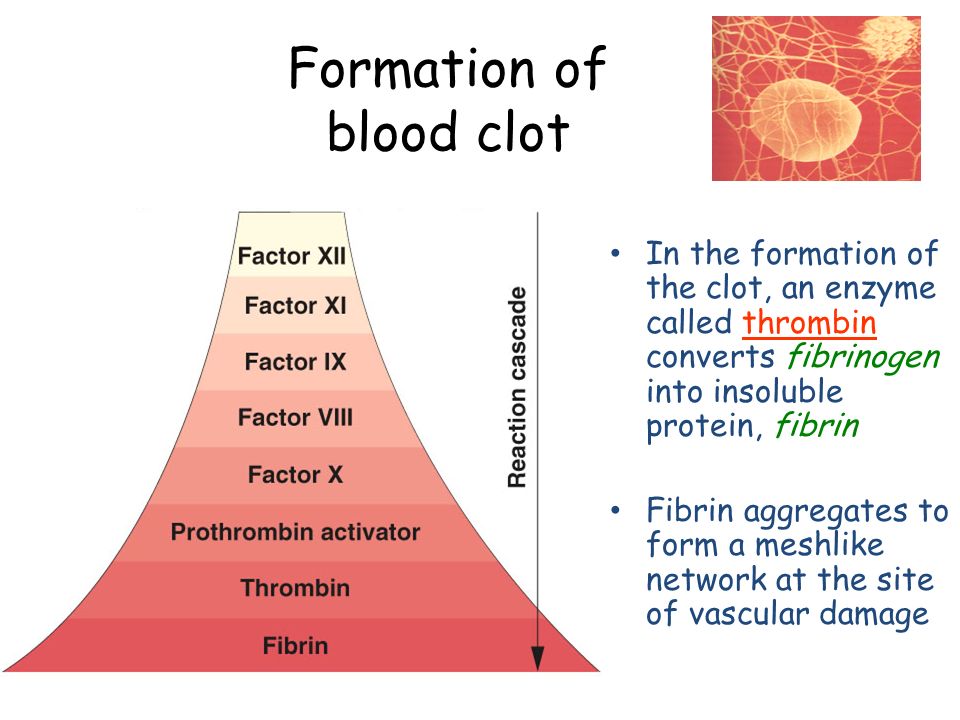
The Transition of Prothrombin to Thrombin
It also predicts that hyponatremia and hypernatremia are risk factors for bleeding and thrombosis, respectively, by favoring the anticoagulant Na+-free and procoagulant Na+-bound forms of thrombin. The two species were found to be approximately equally populated and the distribution between the two forms could be altered predictably by thrombomodulin or by decreasing Na +. This phase begins with the breakdown of the clot. Once bound to the plasma membrane, Factor Xa and Factor Va rapidly associate in a 1:1 stoichiometric ratio to form the prothrombinase complex. Accordingly, this prothrombin variant could be converted to thrombin but failed to yield an active proteinase. Factor Va also decreases the Km of the reaction by enhancing the binding of prothrombin to the prothrombinase complex. This is surprising considering that the area mostly lay fallow for almost 20 years since the numerous studies in the 1980s that established the essential features of prothrombin cleavage by prothrombinase assembled using synthetic phospholipids , , ,.
LOPAP

Both sets of studies implicate a region in the proteinase domain, removed from the active-site of Xa, as contributing to exosite-dependent substrate binding ,. The composite nature of the steady state kinetic constants is illustrated by derivation employing the rapid equilibrium assumption. Two principles emerge that deviate from conventional wisdom that has otherwise dominated thinking in the field. Prothrombinase assembles through reversible interactions between the serine proteinase Xa and the protein cofactor Va on membranes containing phosphatidylserine. Given the relatively weak binding of F12 to thrombin, a significant fraction of the enzyme is unlikely to be regulated in this way.
What is Hemostasis

It is for this reason that prothrombinase rather than Xa is considered the physiological catalyst for rapid thrombin formation following vascular damage. Cleavage in the heavy chain reduces the ability of Factor V to bind to Factor Xa. Using isothermal titration calorimetry to provide very accurate estimates of binding constants, the authors measured binding of F1. As cleavage at two sites is necessary, there are two possible pathways for the formation of thrombin Fig. Indeed, replacement of the P 1 Arg with Gln yielded an uncleavable substrate, but one that bound to prothrombinase with the same affinity as wild type P2.
Chapter 16 Short Answer Questions Flashcards
The external blood clot is also called scab. The Arg 155 site Red Arrow is susceptible to thrombin cleavage and separates the fragment 1 region from fragment 2 within F12. Thus the kinetic constants used to phrase the functional consequences of prothrombinase assembly are based on steady state kinetic constants determined by varying the substrate for the first reaction and measuring the product of the second enzyme catalysed reaction. Instead, mechanistic explanations for some of the observations may lie in the membrane composition of activated cells and hindered diffusion on the crowded cell surface that is sufficient for prothrombinase assembly but sub-optimal for mediating delivery and constrained presentation of substrate to enzyme. Thus, the formation of the prothrombinase complex alters the sequence of prothrombin bond cleavage.
Phys. Exam 2 Hemostasis Flashcards

The first deals with the basis for substrate specificity at the level of the protein substrate: Why does prothrombinase selectively act to cleave prothrombin and not the other structurally related vitamin K-dependent zymogens of coagulation? When oxygen is loaded bound to hemoglobin , the hemoglobin becomes bright red. Sophisticated biophysical and biochemical insights into the players of this reaction were developed in the early days of the field. The intrinsic tenase complex is composed of both and. The platelets which are a play a vital role in it. The extrinsic pathway is faster and gets activated within seconds of tissue damage. How do these transitions in thrombin work? These points illustrate that thrombin occupies a continuum of zymogen-like and proteinase-like forms following initial cleavage. The Question of Substrate Specificity While both cleavage sites are accessible within the substrate to externally added proteinase, prothrombinase essentially exclusively acts at the Arg 320 site when presented with prothrombin.
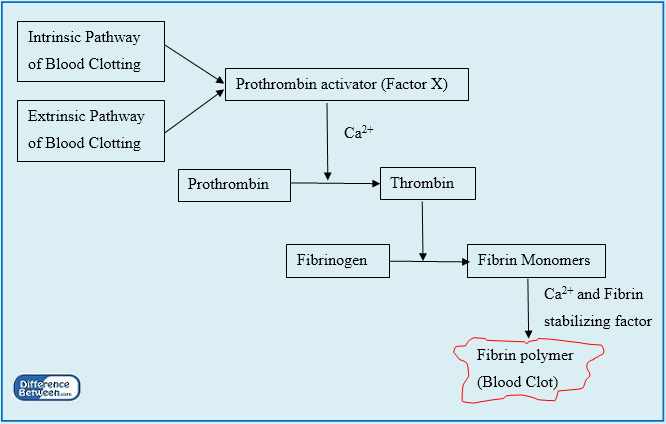
Prothrombin activator

Coupled with the regulation of substrate conformation as a result of the zymogen to proteinase transition, novel mechanistic insights result for numerous aspects of enzyme function. Botrocetin ® is a C-type lectin-like protein isolated from the venom of Bothropps jararaca. Factor V Leiden is an important risk factor for venous thromboembolism, that is, deep vein thrombosis or pulmonary embolism. When thrombin is generated by Factor Xa alone, the first pathway predominates and prothrombin is first cleaved after Arg 271, producing prethrombin 2, which is subsequently cleaved after Arg 320. Prothrombin activator is formed 2. For example, the drug is an indirect inhibitor of Factor Xa. Factor Va strengthens the affinity of Factor Xa for the membrane and also increases the kcat of Factor Xa for prothrombin.
Prothrombinase

Because there are two cleavage events, prothrombin activation can proceed by two pathways. Enzymic specificity at multiple levels comes to the fore in considering the specific action of prothrombinase on prothrombin. The conversion of prothrombin to thrombin is catalyzed by factor Xa and includes two essential proteolytic cleavages, producing three intermediates: prethrombin 2, meizothrombin, and Fragment 1. Early studies performed with Xa in the absence of Va established cleavage at Arg 271 followed by cleavage at Arg 320 as the prominent, or perhaps only, pathway by which prothrombin was activated. Finally, considering the ambiguity in inferring the basis for sequence preference for cleavage by prothrombinase, it seems appropriate to question why the action of the enzyme is restricted to only two sites at the expense of 37 other arginines in the substrate. More recent studies at physiological pH and ionic strength have revealed a much weaker affinity μ m for this interaction indicating that only a modest fraction of thrombin may be bound to F12 in the best case ,. However, despite the fact that Xa readily cleaves peptidyl substrates, its ability to activate prothrombin is markedly low.
What is Hemostasis

What might this test show? It also follows that questions related to how the cofactor may function within prothrombinase and how narrow enzymic specificity towards the protein substrate is achieved represent two sides of the same coin. Provided the P 1 residue was Lys or Arg, the variant substrates were cleaved but with varyingly reduced V max. The initially formed thrombin activates Factor V. Thus, insights drawn from studies with prothrombin are likely to shed new light on a wide swathe of proteolytic reactions related to the blood coagulation response. The specificity of thrombin toward prohemostatic and anticoagulant substrates is accounted for simply by competition between macromolecular ligands: F1. In the most situations, the solid forms out of solute phase, and sink to the bottom of solution though it will float if it is less dense than the solvent or form suspension. Although it has been shown that Factor Xa can activate prothrombin when unassociated with the prothrombinase complex, the rate of thrombin formation is severely decreased under such circumstances.